Research Unit
Degradomics

Prof. Dr. Christoph Becker-Pauly
Tel. +49 431 880-7118
Christoph Becker-Pauly is spokesperson of the DFG-funded Research Training Group ReMPro and has published over 100 research papers on the structure and function of proteolytic enzymes in health and disease. Proteases are considered central nodes in the regulation of the immune system, in neurodegeneration, and in cancer. Among the nearly 600 human proteases, his group is particularly interested in the characterization of metalloproteases, especially meprins, ADAM10/17, and MMPs. Using N-terminomics, cell culture, and mouse models, his group has discovered important biological functions of these enzymes in mucosal homeostasis, Alzheimer's disease, tumor progression, and systemic and local inflammatory diseases.
Research: Prof. Becker-Pauly
Proteolytic enzymes in health and disease
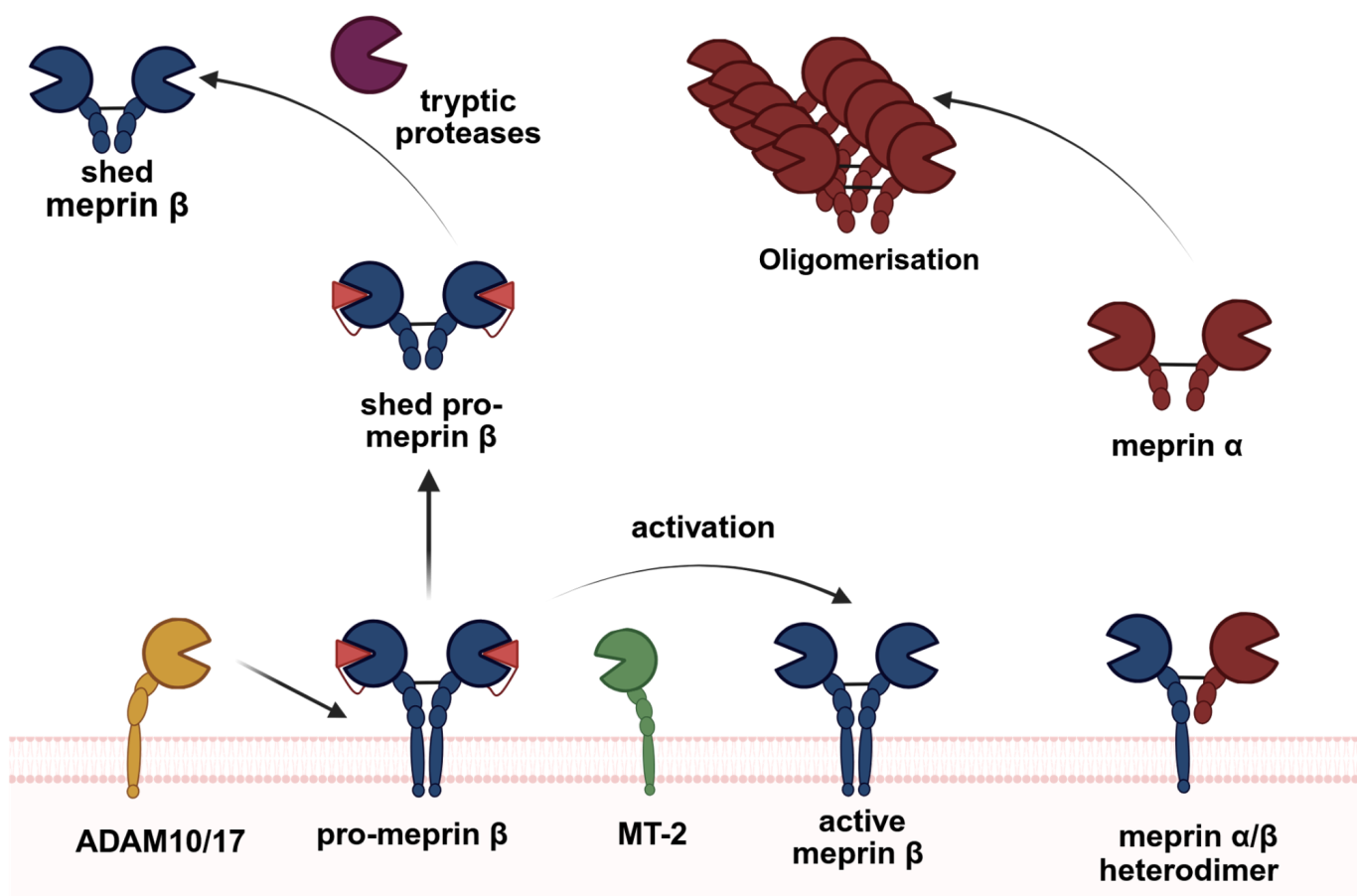
Fig. 1: Regulation of metalloproteases at the cell surface.
Our group is particularly interested in meprin and ADAM metalloproteases and their regulation at the plasma membrane. We identified unique molecular features that impact activity and localization of these enzymes and have consequences for the biological function.
(Arolas et al., PNAS, 2012; Werny et al. FEBS J, 2023; Song et al. Acta Neuropathol, 2024)
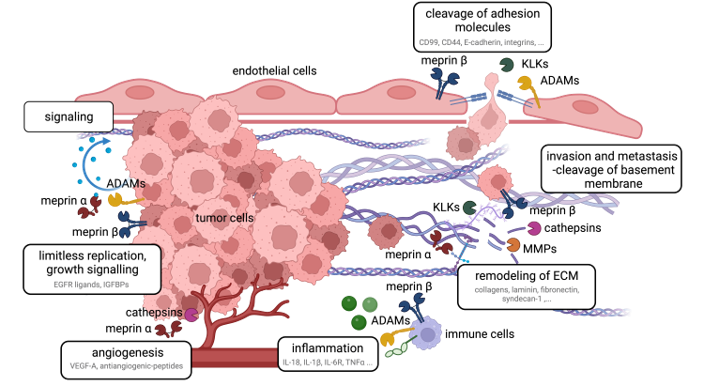
Fig. 2: Tumorprogression and metastasis influenced by proteases and their substrates.
Our aim is to investigate the pathological relevance of these molecules and their molecular properties with regard to potential therapeutic applications.
(Peters & Becker-Pauly, Cancer Metastasis Rev, 2019 ; Bickenbach et al., Sci Adv, 2025)
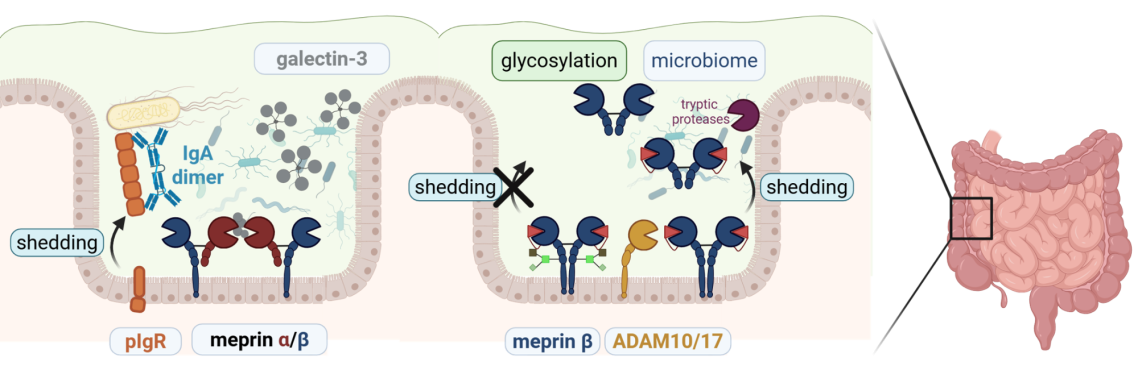
Fig. 3: Host microbiome interaction is modulated by proteolytic enzymes and vice versa.
With the help of mass spectrometry based proteomics approaches, we revealed proteolytic events that regulate the composition of the intestinal microbiome. Importantly, the microbiome itself has great impact on the activity and localization of meprin metalloproteases thereby influencing their immunemodulatory functions.
(Schütte et al., PNAS, 2014; Wichert et al. Cell Rep, 2017; Bülck et al., Sci Adv, 2023)
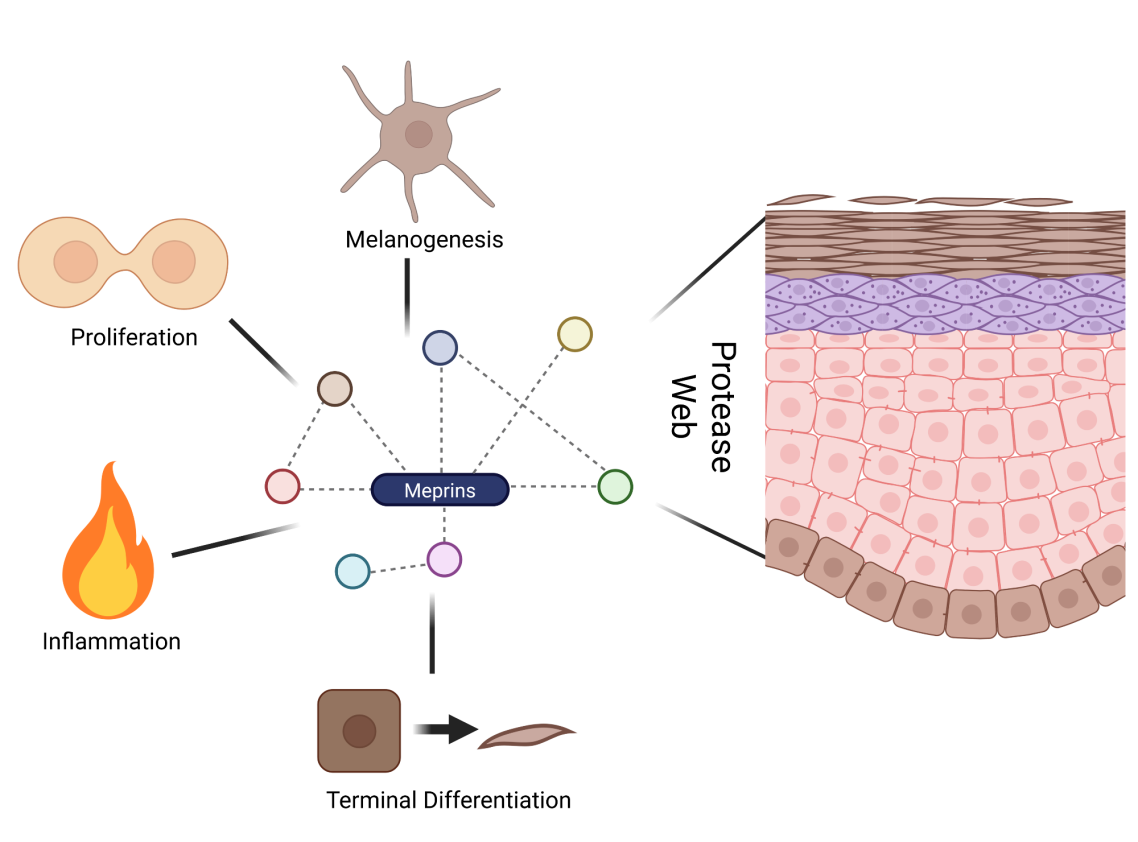
Fig. 4: The proteases web in epidermal homeostasis and beyond.
In different mouse models and human tissue samples, we observed a proteolytic interplay that contributes to keratinocyte differentation and dermal collagen maturation. Current work points toward a systemic immunological function of meprin alpha associated with SIRS.
(Broder et al., PNAS, 2013; Rahn et al. Matrix Biol, 2021; Kruppa et al., Matrix Biol Plus, 2021)
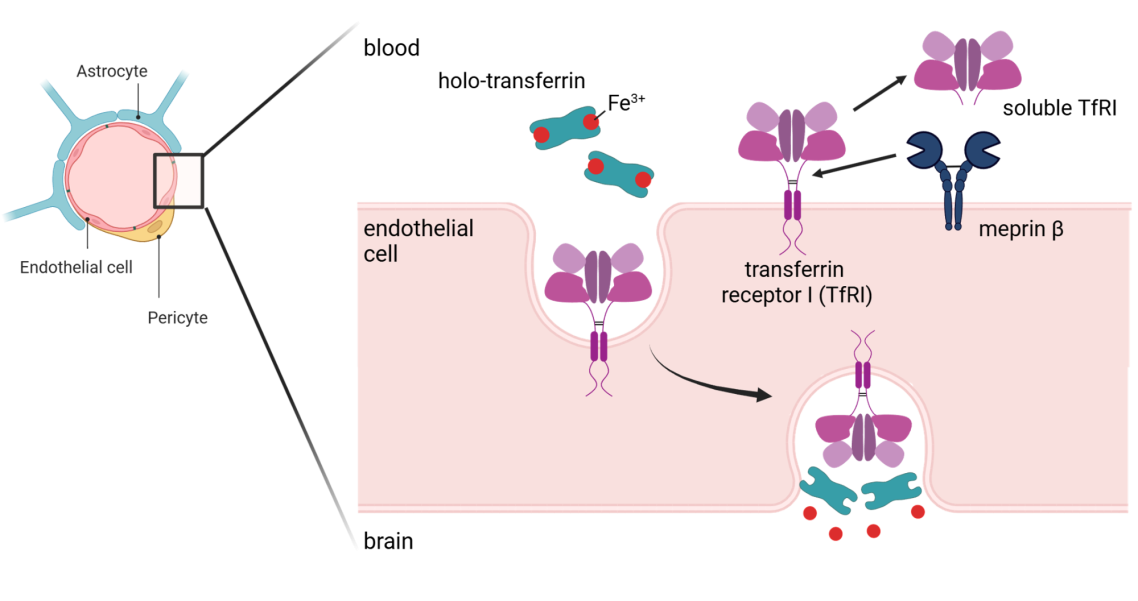
Fig. 5: Meprin β is a key regulator of iron plasma levels.
We and others observed altered iron levels in Mep1b knock-out mice compared to wildtype animals. Employing N-terminomics we identified the transferrin receptor I (TfRI) as a substrate for meprin β, which may be the cause for the different iron concentrations in blood.
