Research Unit
Tumor Biochemistry

Prof. Dr. Elmar Wolf
Tel. +49 431 880-3336
Elmar Wolf is a tumor biologist whose research focuses on oncogenic transcription factors and strategies to therapeutically target them. In collaboration with medicinal chemistry partners, he pursues the approach of directing tumorigenic proteins toward proteasomal degradation using PROTACs (proteolysis-targeting chimeras). These small molecules are not only powerful cell biological tools but also represent an entirely novel and unique therapeutic strategy. Elmar develops PROTACs within projects funded by the ERC (Starting, Consolidator, and Proof of Concept Grants), the German Cancer Aid (consortium project TACTIC), and the DFG (Collaborative Research Center 387).
Research: Tumor Biochemistry (Wolf Lab)
In aging Western societies, cancer represents one of the most pressing medical challenges. A hallmark of tumors is the deregulation of signaling pathways that profoundly alter the transcriptional profile of cancer cells. Transcription factors of the MYC family play a pivotal role in this oncogenic reprogramming.
Our vision is to understand the transcriptional alterations that occur during tumorigenesis—particularly the oncogenic function of MYC—at a level that enables the development of novel pharmacological strategies for cancer therapy. To this end, our group employs genome- and proteome-wide approaches to comprehensively characterize tumor biology, as well as genetic screenings to identify new therapeutic vulnerabilities. In collaboration with our partners, we subsequently develop compounds that interfere with the oncogenic functions of these target proteins. A particular focus lies on the development of so-called PROTACs (Proteolysis Targeting Chimeras), which enable the selective degradation of tumor-promoting proteins. In the coming years, we aim to advance these PROTACs to the stage of clinical testing and, ultimately, therapeutic application.
The oncogenic function of MYC
Almost all human tumors express significantly higher levels of MYC protein compared to normal tissues. Mouse models have further demonstrated a direct link between MYC activity and tumorigenesis. Despite its critical role as an oncogene, MYC was long considered an unsuitable therapeutic target, as it is also indispensable for the function of normal cells. Motivated by the question of whether and how the oncogenic role of MYC differs from its physiological function, we have in recent years investigated the mechanisms of MYC-mediated gene regulation in depth, yielding two key insights.
First, we demonstrated that MYC binds to the promoters of all expressed genes and can regulate them (Walz et al., Nature, 2014). However, target genes differ markedly in their affinity for MYC (Lorenzin et al., eLife, 2016). High-affinity targets are regulated even at low MYC concentrations, as found in normal cells, and are essential for cell growth and proliferation. At the very high concentrations present in tumor cells, MYC additionally binds to low-affinity promoters, thereby activating genes that drive tumor-specific processes.
Second, our data support the unexpected model that MYC does not primarily regulate initiation - that is, the transcription rate - but rather loads RNA polymerase II with elongation factors. In the absence of this loading mechanism, RNA polymerase loses much of its processivity (Baluapuri et al., Molecular Cell, 2019; Vogt et al., Gut, 2024). Moreover, MYC also exerts transcription-independent functions, such as preventing DNA damage through its multimerization (Solvie et al., Nature, 2022).
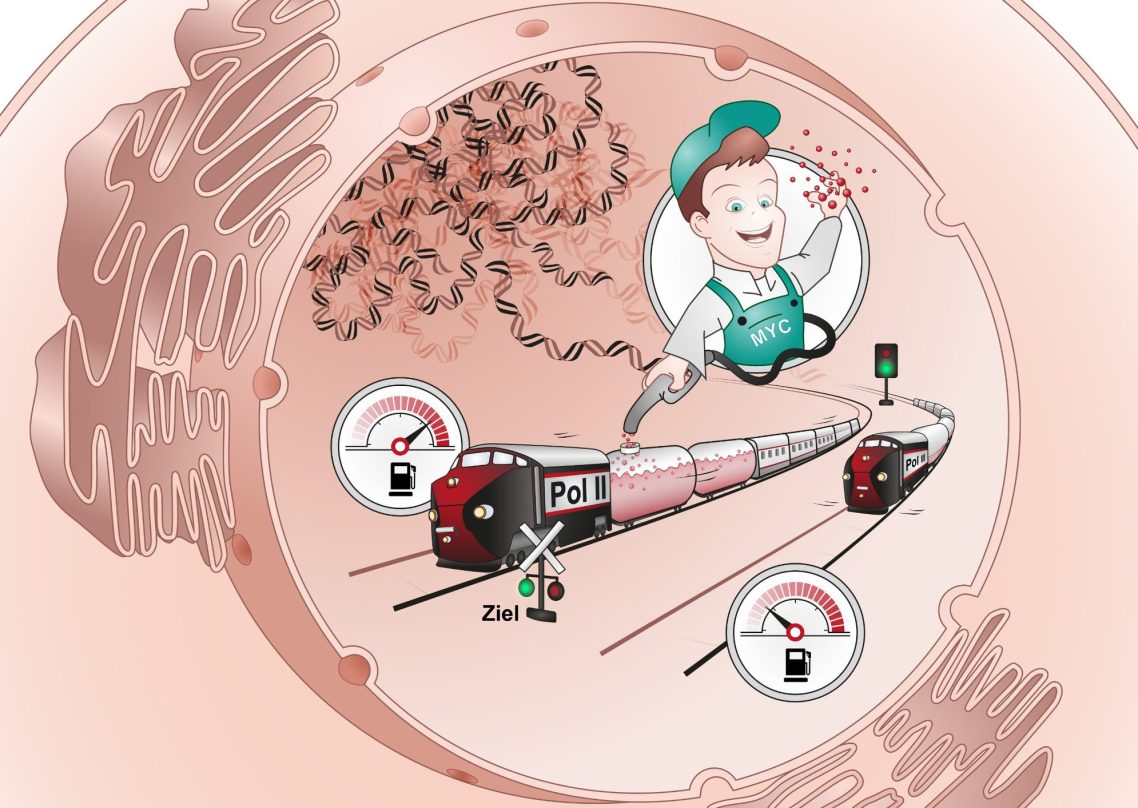
Investigating transcriptional elongation
Our observation that MYC regulates not transcriptional initiation but the elongation behavior of RNA polymerase II sparked our broader interest in this fundamental cellular process. To dissect elongation in detail, we developed and applied methods such as mass spectrometry, 4sU-sequencing, and ChIP-sequencing. These approaches enabled us to identify several factors with critical roles in elongation. The histone chaperone SPT6 directly supports the processivity of RNA polymerase (Narain et al., Molecular Cell, 2021), while SCAF1 ensures efficient elongation through GC-rich genomic regions (Bhandare et al., Nucleic Acids Research, 2025).
New pharmaceutical concepts in oncology
Our findings not only advance fundamental models of MYC function and transcriptional regulation but also provide new opportunities for the development of innovative therapeutic strategies. For many years, we have been developing PROTACs targeting MYC-interacting proteins. PROTACs are bifunctional small molecules that recruit E3 ubiquitin ligases into proximity with their target proteins, leading to ubiquitination and subsequent proteasomal degradation.
We have successfully developed PROTACs against AURORA A and B (Adhikari et al., Nature Chem Bio, 2020; Bozilovic et al., Current Research in Chemical Biology, 2022; Sflakidou et al., ACS Pharmacology, 2024) as well as against WDR5 (Dölle et al., JMC, 2021). In addition, we have established assays that allow the prediction of the suitability of specific E3 ligases for defined target proteins.


